|
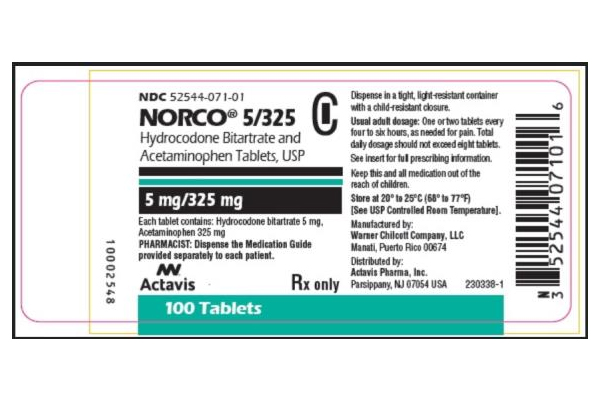
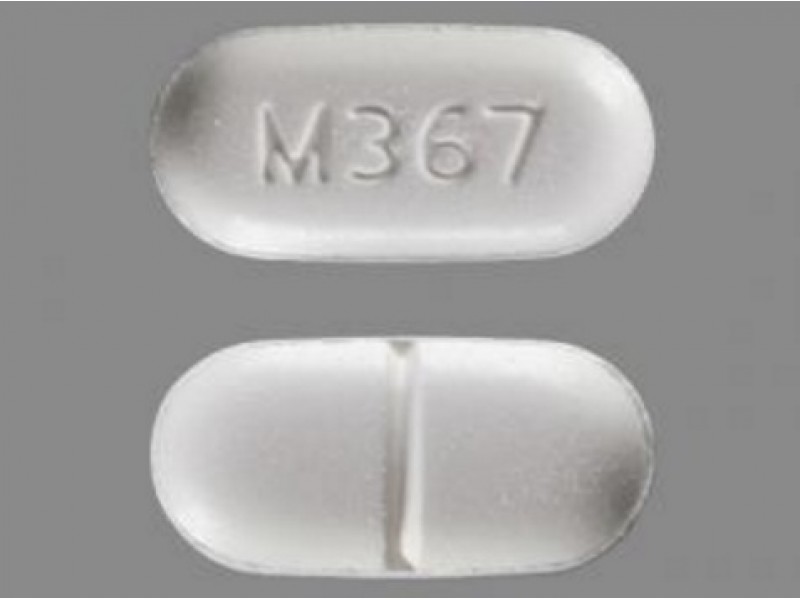
The subject has an ongoing abuse of illicit substances, alcohol, or actively smoking marijuana.Previous participation in a clinical research trial within 30 days prior to randomization.Individuals who are cognitively impaired or who are not able to give informed consent.Health concerns that the study physician feels may confound study results.Subjects who are taking concomitant medications or Nutraceuticals that interfere with Hydrocodone metabolism as listed in Appendix 11 and/or as deemed clinically significant by a pharmacovigilance team that is contracted to monitor and advise.Subjects must have signed an informed consent document indicating that they understand the purpose of and procedures required for the study and are willing to participate in the study.Subjects currently on hydrocodone/APAP must be taking minimal daily dose of 15mg of Hydrocodone for at least 30 days.Have been taking hydrocodone/APAP for their chronic non-cancer pain. Norco (hydrocodone bitartrate and acetaminophen) is an opioid analgesic and antitussive (cough suppressant) combined with a pain reliever (analgesic) used to treat moderate to fairly severe pain.Because Norco contains the narcotic hydrocodone, it can be addictive and should be used with caution. It is intended to relieve moderate to severe pain. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
